Serevent

Serevent
- Type: Control
- Manufacturer: GSK
- Substance: Salmeterol
- Dosage: 25 mcg
- Avg Price: $382
Pharmachologic Effect
Serevent is indicated as a bronchodilating drug. It contains salmeterol, which has a prolonging effect and does not have any influence on the cardiovascular system when used in therapeutic doses. The drug effect lasts up to 12 hours. The continuous use of salmeterol helps to improve the functions of lungs, to reduce obstructing syndrome and night bronchoconstriction among patients with bronchial asthma.
The majority of patients with asthma do not need additional use of bronchodilators if they regularly take Serevent aerosol; however such patients should always bring with them short-effect bronchodilators, taking into consideration the fact that the drug Serevent is not used to relief acute episodes of bronchospasms.
Indication
Serevent is used for regular and continuous treatment of the patients, who have a reversible obstruction of respiratory system, which is associated with bronchial asthma, including night attacks, also bronchoconstriction, associated with physical activity.
The drug Serevent can be prescribed to the patients with the chronic forms of obstructive lungs diseases.
Application Method
Serevent is intended only for inhalation use. The aerosol should be taken regularly. The drug is not effective for cupping the acute bronchospasm.
Over The Counter Serevent
Serevent Otc is not available now. You can buy Serevent at pharmacy onliny having valid prescription. Nevertheless, you can always order Generic Serevent at online pharmacy. Generic Serevent is significantly cheaper that the brand one, and the prescription is not needed when oredering it online.
Dosage
The dose of the drug Serevent, the scheme of therapy, as well as additional treatment is prescribed by a specialist. During the treatment of patients with bronchial asthma, it is necessary to control the general condition of the patients regularly, as well as their function of the lungs.
Adult patients with bronchial asthma and chronic lungs diseases which are followed by an obstructive syndrome are usually prescribed 2 inhalations of Serevent two times a day. Patients with a hard obstruction of a respiratory system and also when the doses mentioned above are not effective the dose of salmeterol can be increased up to 4 inhalations two times a day. Self-correction of the dose of Serevent as well as finishing of the treatment or changing of the doses of the drug are unacceptable.
Patients who are older than 4 years are usually prescribed 2 inhalations of the drug Serevent twice a day.
Patients under 4 years should not be prescribed Serevent (due to the lack of clinical testing of salmeterol in this age group of patients).
Older patients, as well as those with kidney failure, do not need any correction of the dose of salmeterol.
The Use Of The Serevent Aerosol
The specialist should show the patient the technique of using the Serevent aerosol, and also check the correctness of the use. If the aerosol Serevent is used incorrectly, the amount of active substance entering the lungs decreases, which may lead to a decrease in the effectiveness of bronchodilating action. Little children may need the help of adults during the inhalation.
It’s necessary to clean the plastic case of Serevent aerosol regularly, not less than once a week, wiping it with a dry napkin. Do not remove the balloon from the plastic body, or put the product into the water.
Warning
During the therapy with the drug Serevent there is a possibility of development of undesirable symptoms, including tremor, headache, palpitation, dizziness. As a rule, these undesirable phenomena are reversible, they decrease or disappear completely when the treatment is continued or the dose of the drug is reduced.
Some patients during the use of Serevent aerosol face the development of the heart rhythm disturbance, including atrial fibrillation, extrasystole and tachycardia. These effects developed rarely, the probability of occurrence of such effects is higher within the patients with the cardiovascular system, as well as during the use of high doses of Serevent aerosol.
As with the use of other inhaled drugs, during the treatment with Serevent aerosol, some patients reported the development of bronchospasm. In such cases, it is necessary to stop using Serevent aerosol immediately and prescribe fast-acting bronchodilator drugs to the patients and, according to the specialist's decision, another alternative therapy.
In rare cases, during the use of salmeterol , the joint pain, muscle cramps, reactions of hypersensitivity, including skin rash, angioedema and anaphylactic shock were recorded.
Serevent Aerosol can lead to the development of local negative phenomena, irritation of the mucous membrane of the pharynx and oral cavity.
Side Effects
From The Respiratory System
The development of paradoxical bronchospasm is possible (in this case, immediately cancel the drug and prescribe an alternative therapy)
From The Cardiovascular System
Possible resctions: headache, tachycardia, (it should be borne in mind that these symptoms may not be associated with taking the drug); in predisposed patients - cardiac arrhythmias (ciliary arrhythmia, supraventricular tachycardia and extrasystole).
Pregnancy And Breastfeeding
The use of the drug during pregnancy and lactation (breastfeeding) is possible only if the intended benefit to the mother exceeds the potential risk to the fetus or baby.
Comparison
- Serevent vs Budesonide
- Serevent vs Albuterol
- Serevent vs Spiriva
- Serevent vs Proventil
- Serevent vs Advair
- Serevent vs ProAir
- Serevent vs Atrovent
- Serevent vs Primatene
- Serevent vs Foradil
- Serevent vs Qvar
- Serevent vs Levalbuterol
- Serevent vs Ipratropium
- Serevent vs Wixela
- Serevent vs Tiotropium
- Serevent vs Epinephrine
- Serevent vs Ciclesonide
- Serevent vs Trelegy
See Also
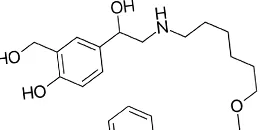
Salmeterol
Salmeterol is a prescription drug for the patients 4 years of age or older. It is a medication intended to fight the symptoms of asthma and COPD. It is produced in a powder form, which works by relaxing the muscles and expanding the airways. The drug is often used to suppress or prevent bronchospasm.

